Developmental constraints
Developmental constraints shape the evolution of the nematode mid-developmental transition.
Zalts H and Yanai I. Nature Ecology and Evolution 2017 doi:10.1038/s41559-017-0113
Since the works of Ernst Haeckel and Karl Ernst von Baer in the 19th century, it has been recognized that a stage in mid-embryogenesis is remarkably conserved across vastly different vertebrates such as fish, amphibians, and mammals. The underlying mechanism, however, which sustained this apparent conservation, has been one of the greatest mysteries of the field of developmental biology. Indeed, the difficulty of this problem even led many researchers to conclude that this stage – known as the phylotypic stage – was an illusion, present only in the mind of the biologist and of no consequence for the organism. With the discovery of HOX genes, though, and their unique expression during this stage, breakthrough works in evolutionary developmental biology greatly expanded evidence supporting the phylotypic stage. A consensus has been emerging over the past three decades following the work of Duboule and Raff that the phylotypic stage is constrained by the crucial expression of these genes, no evidence has been provided for the actual mode of evolution of this stage.
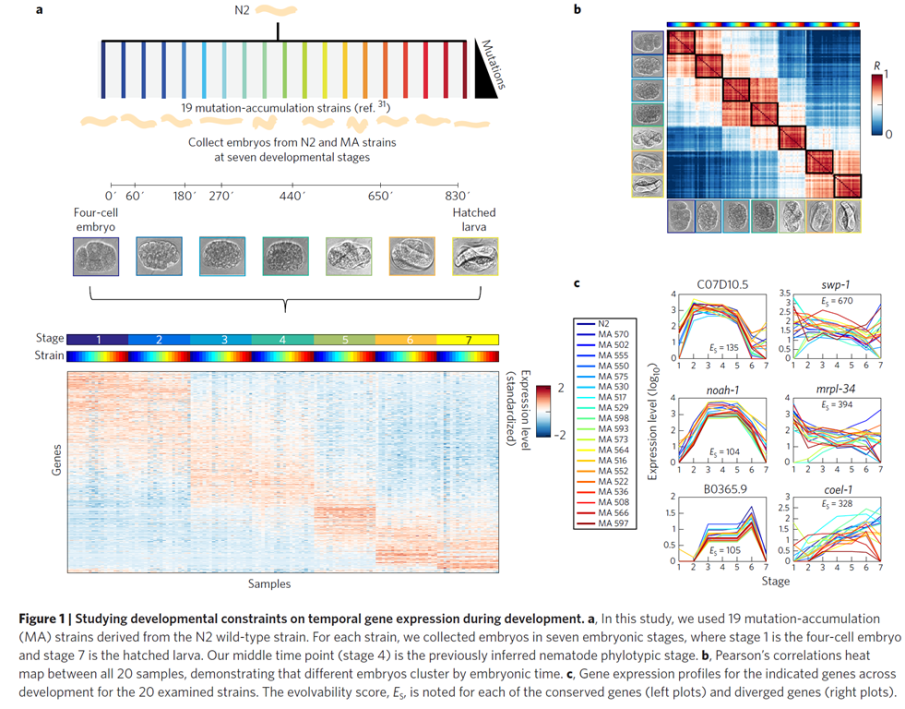
In our new manuscript, we provide the first molecular evidence that the phylotypic stage is under developmental constraints relative to other embryonic stages. We achieved this by framing the problem in the ultimate locus of evolution – the population. We compared actual populations with standing variation using a novel population-genetic approach to testing for developmental constraints. We compared twenty strains that have accumulated changes in the absence of positive selection for 250 generations by collecting thousands of individual embryos and queried their transcriptomes across seven developmental stages producing a unique and vast dataset. Analyzing it, we found uneven distributions of variation across developmental time, across functional gene groups, and germ-layer specific genes. Together our results provide the first experimental evidence for developmental constraints in development: in the absence of positive selection, the phylotypic stage is slower to accumulate change relative to other stages.
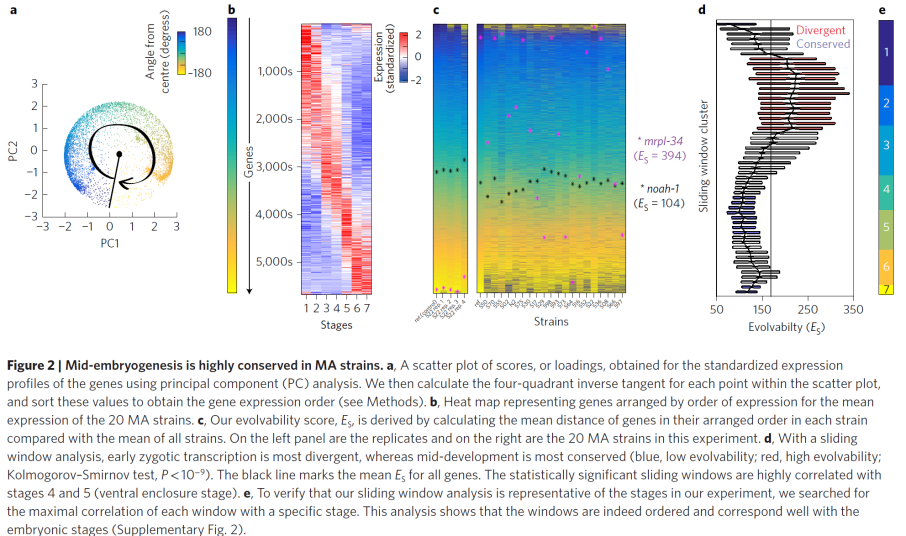
We believe that our work sets up a new population-level study of developmental constraints, however it also opens up more questions than it answers: How do embryonic stages emerge? Given developmental constraints on stages, how can new species evolve? How are the developmental constraints manifested in cells of different cell-types? The approach we follow here will undoubtedly also be useful to others asking such questions relating to selective pressures on developmental properties such as aging, developmental novelties, and disease.
evolution papers developmental biology evolution gene expression








